- Main
- Engineering - Engineering - General & Miscellaneous
- ISO 13485: 2016: A Complete Guide to...

ISO 13485: 2016: A Complete Guide to Quality Management in the Medical Device Industry, Second Edition
Itay AbuhavSukakah anda buku ini?
Bagaimana kualiti fail ini?
Muat turun buku untuk menilai kualitinya
Bagaimana kualiti fail yang dimuat turun?
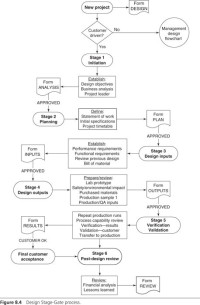
This book will be a substantial revision, which will reflect the new version of the ISO 13485:2016. This represents the standard protocols that all medical device manufacturers must follow, in the fabrication of their products. It will focus on changes in the structure of the quality management system; change in the documentation for quality managemeent systems and finally, present the different methods of implementation of the standard requirements within the organization.This new version was initiated in 2016, thus all apprpriate enterprises using the old standard must convert to the new version, now available.The Second Edition will clarify, explain and demonstrate the new version.
Tahun:
2018
Edisi:
Second edition
Penerbit:
CRC Press
Bahasa:
english
Halaman:
878
ISBN 10:
1351000799
ISBN 13:
9781351000796
Fail:
PDF, 105.67 MB
Tag anda:
IPFS:
CID , CID Blake2b
english, 2018
Baca dalam Talian
- Memuat turun
- pdf 105.67 MB Current page
- Checking other formats...
- Menukar menjadi
- Unlock conversion of files larger than 8 MBPremium
Selama 1-5 menit fail akan dihantar ke e-mel anda.
Dalam masa 1-5 minit fail akan dihantar ke akaun Telegram anda.
Perhatian: Pastikan bahawa anda telah memautkan akaun anda kepada bot Telegram Z-Library.
Dalam masa 1-5 minit fail akan dihantar ke peranti Kindle anda.
Harap maklum: anda perlu mengesahkan setiap buku yang ingin dihantar ke Kindle anda. Semak e-mel anda untuk pasti ada e-mel pengesahan dari Amazon Kindle Support.
Penukaran menjadi sedang dijalankan
Penukaran menjadi gagal
Faedah Status Premium
- Menghantar ke pembaca elektronik
- Peningkatan had muat turun
 Tukar fail
Tukar fail Lebih banyak hasil carian
Lebih banyak hasil carian Faedah lain
Faedah lain
Istilah utama
Koleksi pilihan



































































































 Amazon
Amazon  Barnes & Noble
Barnes & Noble  Bookshop.org
Bookshop.org